
Seamless Integration Across the Supply Chain
Working and coordinating with several providers can be demanding and time consuming when outsourcing a project.
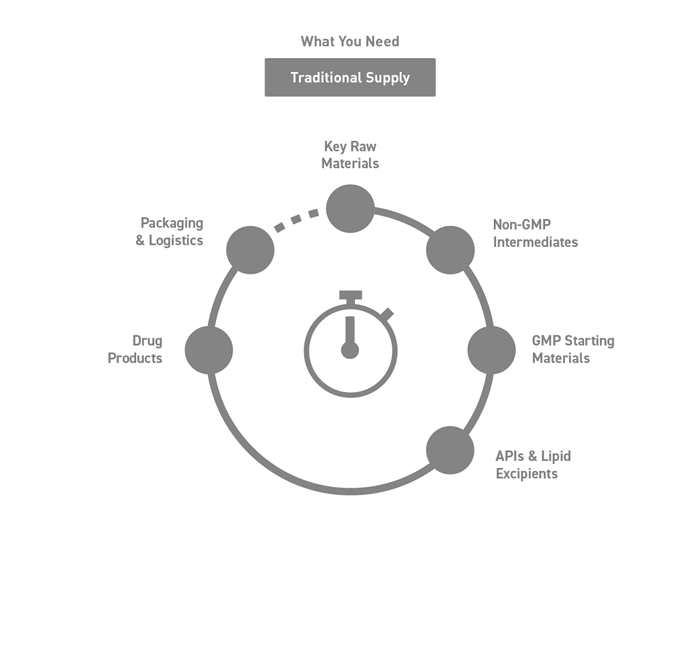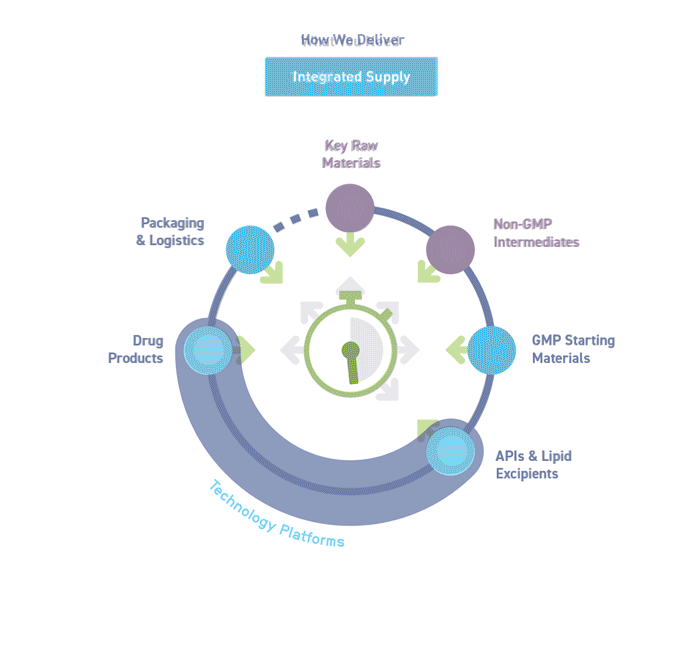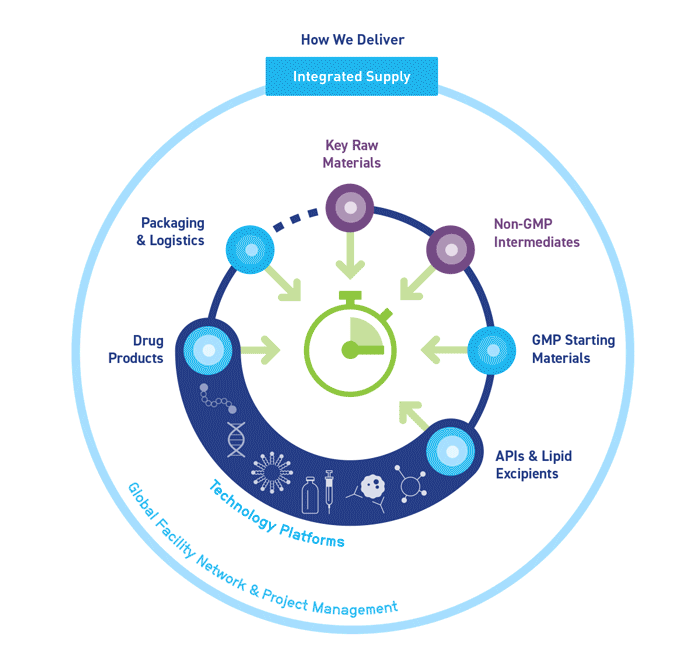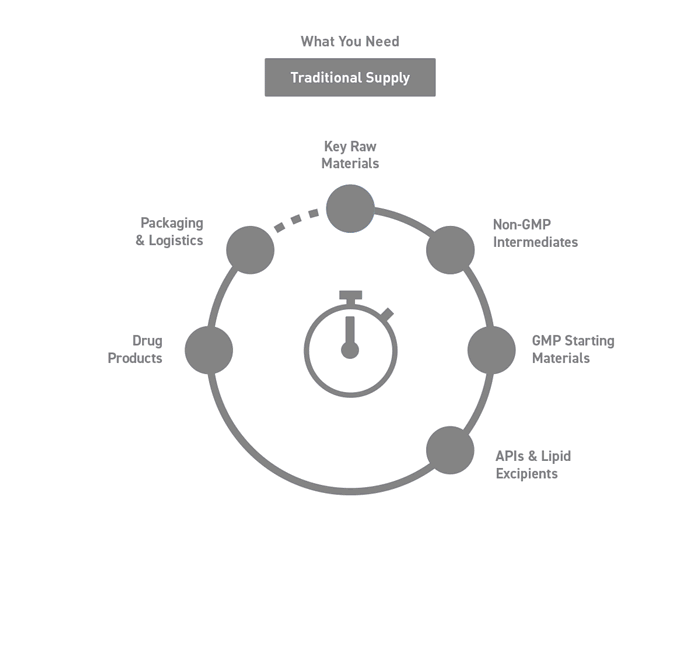
Our aim is to streamline your process, improve efficiency and reduce costs by consolidating multiple services in one organization.
We are the CDMO partner you need to understand and anticipate your project needs. Through seamlessly integrated technologies and expert services across our global network, we support the development and manufacture of your complex modalities.
Our bespoke end-to-end solutions span your supply chain, covering all stages from drug development to commercialization.
Want to find out more?
Get in touch with our team of experts to explore bespoke end-to-end CDMO support of your complete drug lifecycle
at any scale.

